Not signed up to our monthly newsletter? You can sign up for free here.
Echocardiography is a skill that takes a lot of time and practice, but you don’t have to dedicate hundreds of hours to it in order to rule serious cardiac pathology in or out; nor do you necessarily need a cardiac-grade machine or a phased array transducer. In the article below, I discuss ways you can screen for major causes of heart murmurs, breathlessness or syncope without the use of spectral Doppler. All of these techniques can be performed with a microconvex probe.
One of the most common indications for a cardiac scan is the discovery of a murmur. In dogs, the most common cause is mitral valve disease; in cats, either obstruction or regurgitation as a result of hypertrophic cardiomyopathy (HCM).
Hearing a heart murmur in a feline patient can strike fear into any vet working the animal up for even a routine operation, such as a spay. The big worry is, of course, HCM, where a change in loading conditions under anaesthetic could lead to new or worsening outflow tract obstruction, mitral regurgitation, or ischaemia. Severe HCM and associated diastolic dysfunction and dilated left atrium can result in thrombus formation, particularly in the left atrial auricle.
Above: Example of a thrombus in the left atrial auricle (or appendage), seen with intraoperative transosophageal echocardiography using 2D biplane, and then 3D.
While Doppler assessment of diastolic function and myocardial velocities are an integral part of the cardiologist’s repertoire, these tools are not necessary for a screening protocol designed to pick up animals with more advanced (and therefore overt) disease.
Above: Right parasternal long-axis view, demonstrating increased wall thickness which can be appreciated by eye.
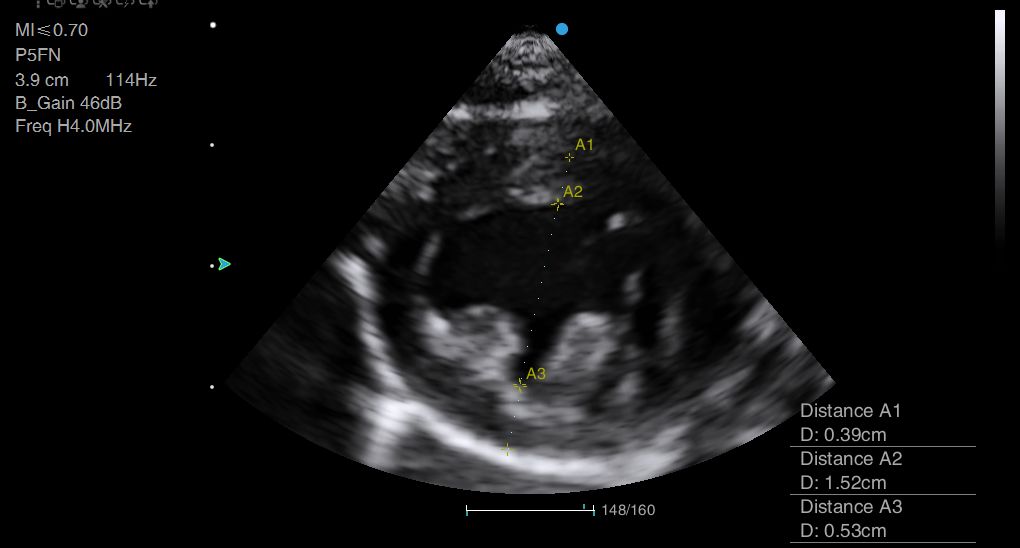
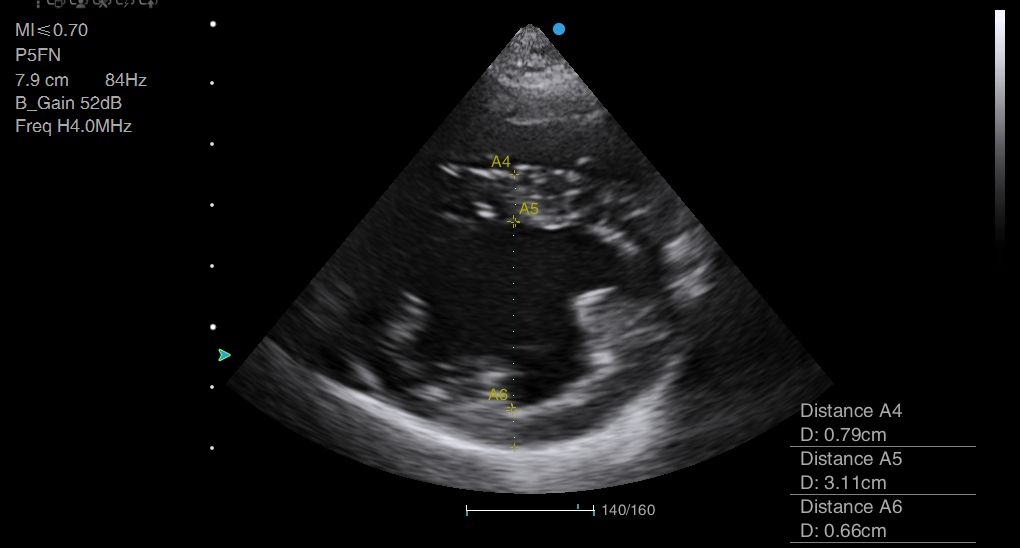
Above: Examples of measuring wall thickness and diastolic cavity diameter from the short axis in a dog.
Severe HCM is defined as wall thickness >9mm (Payne et al., 2013). There is disagreement over whether a 5mm or 6mm wall thickness at end diastole should be the cut-off for a positive diagnosis of HCM (Silva et al., 2013). If you are simply performing a screening test, you may decide that you do not require such high sensitivity and feel it is acceptable to accept the higher value of 6mm. After all, you are probably not looking to ‘diagnose’ HCM, but simply to gauge anaesthetic risk.
More recent studies (Schober, Savino and Yildez, 2017; Häggström et al., 2016) have recommended adjusting for body weight using allometric scaling. It is worth discussing the current literature and deciding, as a practice, where you wish to set your tolerances. Also bear in mind that the distribution of hypertrophy is rarely purely concentric, with the septum often the thickest part. The biggest pitfall when measuring septal thickness is to accidentally include parts of the right ventricular chordae, papillary muscles or moderator band.
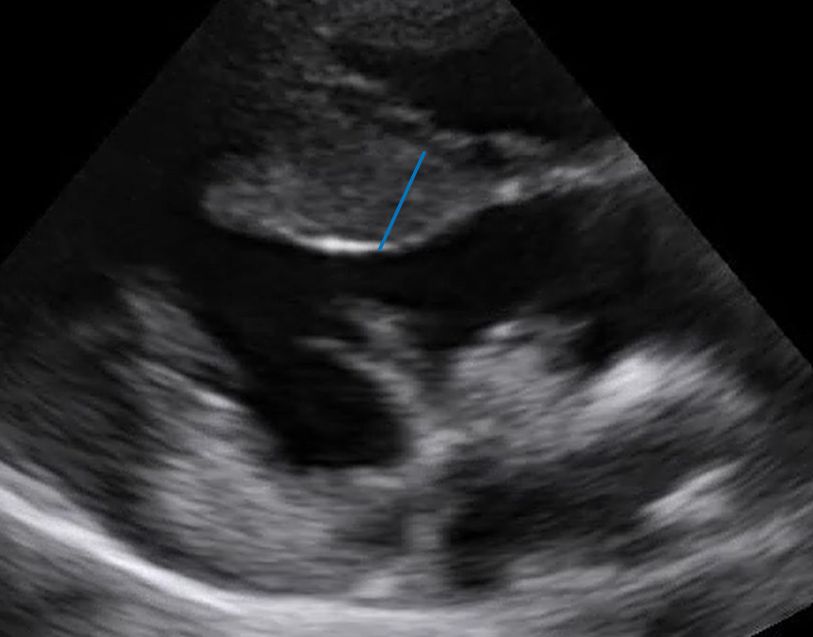
Above: The true septal width is marked in blue. Notice how chords in the right ventricle run along the top of it, and could easily be erroneously included in the septal measurement.

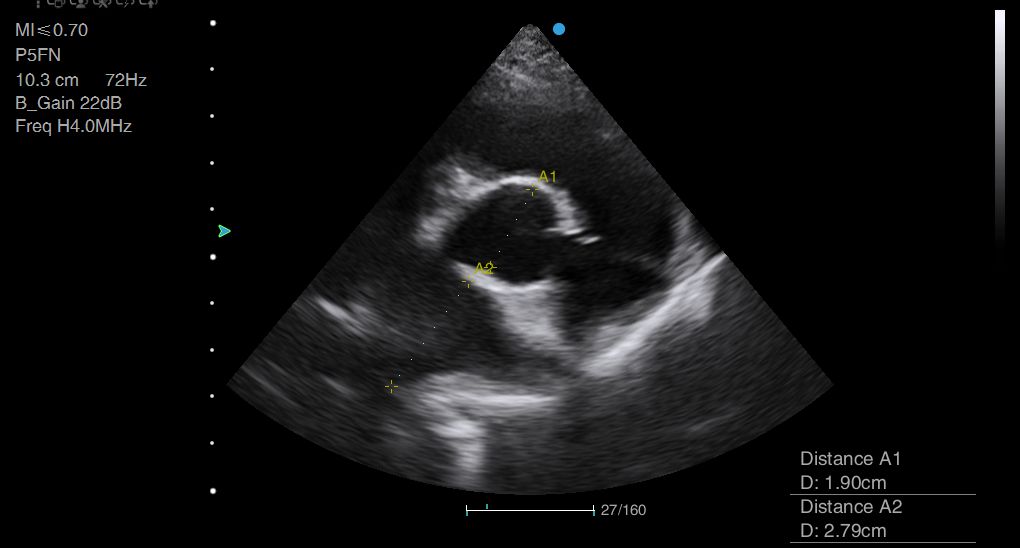
Above: Two examples of LA:Ao ratio measured in a dog. This should be measured when the aortic valve leaflets are closed in both dogs and cats.
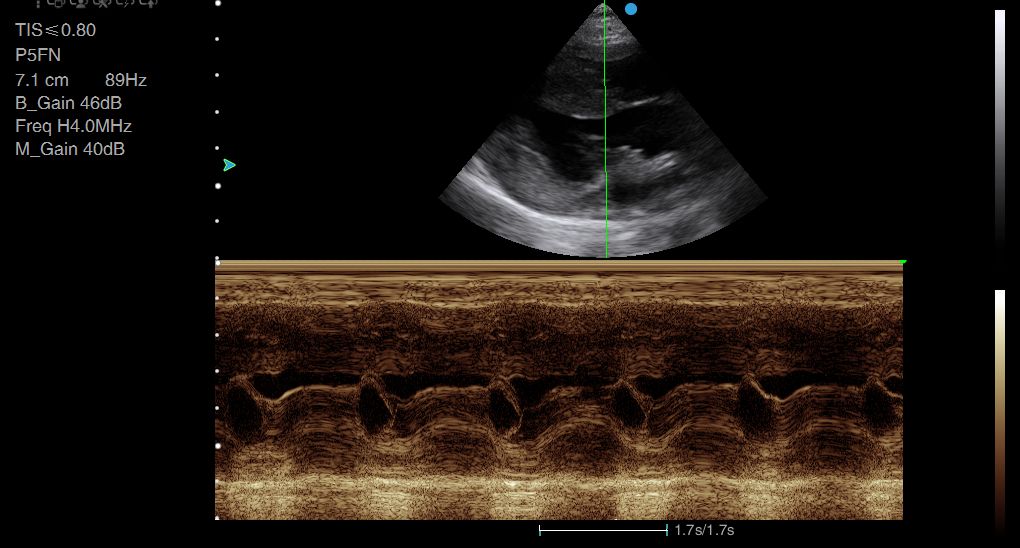
Above: Example of M-mode.
Other echocardiographic signs of more advanced HCM include impaired systolic function and pulmonary hypertension, but for a feline screening test, it is sufficient to become proficient at using wall thickness and left atrial size to rule significant disease in or out.
The most common cause of a murmur in dogs is mitral valve disease. This is usually primary (i.e. a disease of the valve itself), but in dogs with dilated cardiomyopathy (DCM), mitral regurgitation (MR) can be secondary to ventricular dilatation.
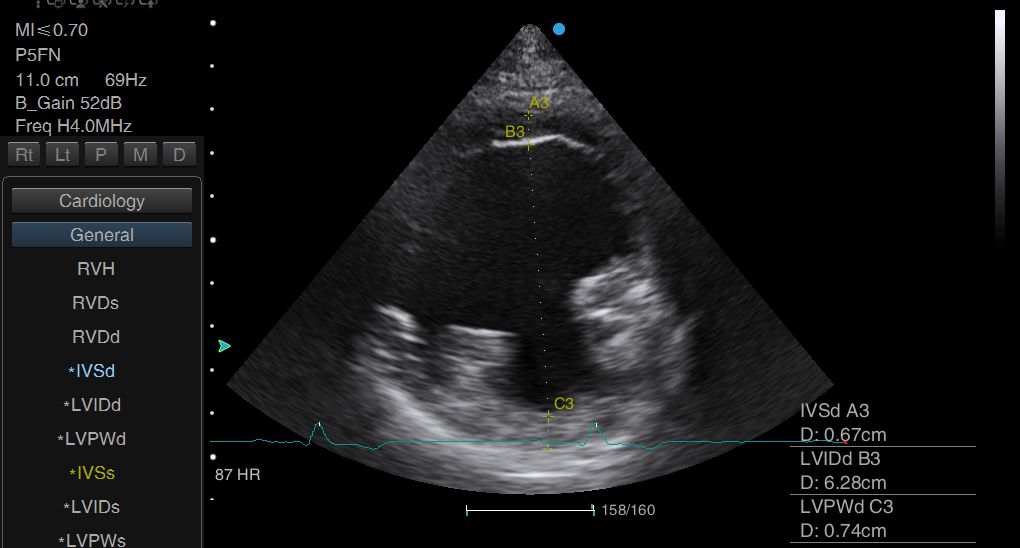
Above: In this dog with DCM, the ventricle measures 6.3cm in diastole.
If your machine has high quality colour Doppler, you can screen for MR very quickly. If your colour frame rates are too low or the colour is prone to ‘bleeding’ and not giving you a lot of useful information, do not despair; there are plenty of 2D signs you can also look out for.
Above: Severe mitral regurgitation is obvious with high quality colour Doppler, as seen here with the Siui Apogee 2300.

Above: Even from 2D imaging, it is clear that the two leaflets do not meet in systole and there is a visible gap. Also notice how obviously the left atrium is dilated.
Above: Dilated right ventricle in a cat (on the left of the screen), which dwarfs the left ventricle.
Besides preoperative screening in the presence of murmurs, the other major indication for a cardiac screen are acute admissions. Classic cardiac symptoms include breathlessness and syncope, and you may wish to be able to quickly rule a major cardiac cause in or out right away. Valve disease is another common cause of these symptoms, but a more acute cause can be pericardial effusion. A quick screen can be potentially life-saving in the case of impending cardiac tamponade, and we have dedicated an entire article to pericardial vrs pleural effusions here.
Don’t believe that this can all be done with a lower end machine? Check out cardiac screening with the Scan Pad – a £2800 machine!
References
Bonagura JD. 2000. Feline echocardiography. J Feline Med Surg 2, 147-151.
Häggström, J., Andersson, Å., Falk, T. et al. (2016). Effect of Body Weight on Echocardiographic Measurements in 19,866 Pure‐Bred Cats with or without Heart Disease. J Vet Intern Med. 2016 Sep-Oct; 30(5): 1601–1611.
MacLea, H., Boon, J., Bright, J. 2013. Doppler echocardiographic evaluation of midventricular obstruction in cats with hypertrophic cardiomyopathy. J Vet Intern Med. Nov-Dec;27(6):1416-20.
Payne, J., Borgeat, D., Connolly, A., et al. 2013. Prognostic Indicators in Cats with Hypertrophic Cardiomyopathy. J Vet Intern Med;27:1427–1436.
Schober, K., Savino, S., Vedat, Y. 2017. Reference intervals and allometric scaling of two-dimensional echocardiographic measurements in 150 healthy cats. J Vet Med Sci. 79(11): 1764–1771.
Silva, A., Muzzi, R., Oberlender, G., et al. 2013. Feline hypertrophic cardiomyopathy: an echocardiographic approach. Arch Med Vet 45, 1-6.
© 2025 | AUA – All Rights Reserved
